COUNTDOWN DAY 1: Interdisciplinary Collaboration
When it comes to MDS assessments, teamwork makes the dream work. The MDS is a critical component of comprehensive resident care in long-term care facilities. Completing these assessments effectively requires an interdisciplinary team (IDT) approach. Interdisciplinary collaboration in MDS streamlines the assessment process and has the power to elevate the quality of care delivered to residents.
IDT meetings are the cornerstone of effective interdisciplinary collaboration, and the frequency of these meetings should be carefully considered. Regular meetings provide a platform for team members to share insights, discuss resident cases, and align on care plans.
Effective communication is at the heart of interdisciplinary collaboration. To create meaningful plans of care, it is crucial to communicate with various IDT members. This includes residents themselves, other disciplines who have recently interacted with the resident, direct staff from all shifts, and the resident’s physician. Each of these sources can provide valuable insights into the resident’s condition, preferences, and progress. By involving multiple disciplines, care plans can be tailored to align with the resident’s unique preferences and goals. This ensures that residents not only receive medically necessary care, but also maintain their dignity and quality of life.
Achieving success in MDS assessments is a collective effort. By fostering interdisciplinary collaboration, long-term care facilities can navigate the evolving landscape of MDS assessments while continuing to deliver high-quality, resident-centered care. Collaboration truly is the key to unlocking success in resident assessments and care planning.
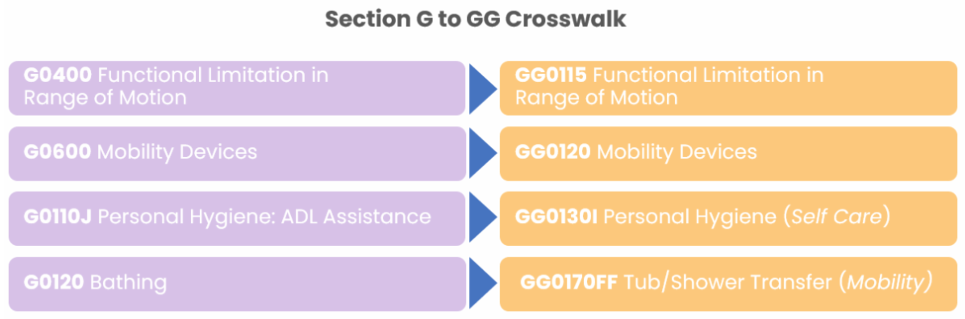
Reliant’s Interdisciplinary Team Meeting Clinical Resource
IN CASE YOU MISSED IT